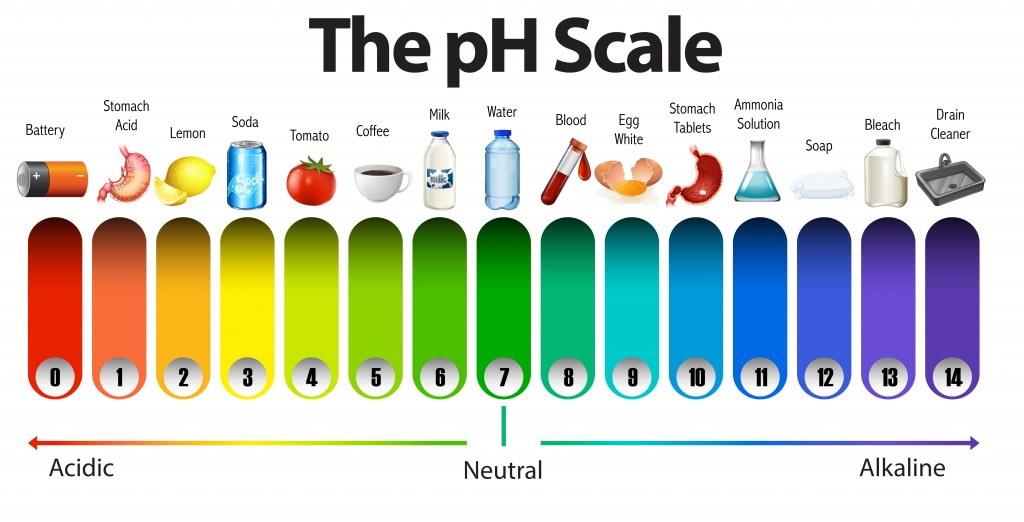
The pH of water is a metal commonly found in natural water supplies. The pH scale is a logarithmic scale ranging from 0 to 14, where a pH of 7.0 is considered to be neutral. Any water with a pH lower than 7.0 is said to be “acidic” and water above 7.0 is called “basic”, as shown in the pH scale below.
Most natural rainwater is acidic, with pH levels often approaching 5.0 – 6.0. This is because rainwater is very low in dissolved solids, and the nearly pure H2O reacts easily with CO2 in the air to form carbonic acid. As rainwater enters the groundwater system it dissolves minerals from the soils and rocks, adding hardness and raising the pH.
The primary problem with acidic or corrosive water is that it tends to dissolve metal components in plumbing systems and water-using appliances. Homes with copper plumbing and metal fixtures are especially susceptible to this problem. The most common sign of low pH is blue-green staining on porcelain bathroom fixtures, showers, toilet bowels, or a metallic taste to the water.